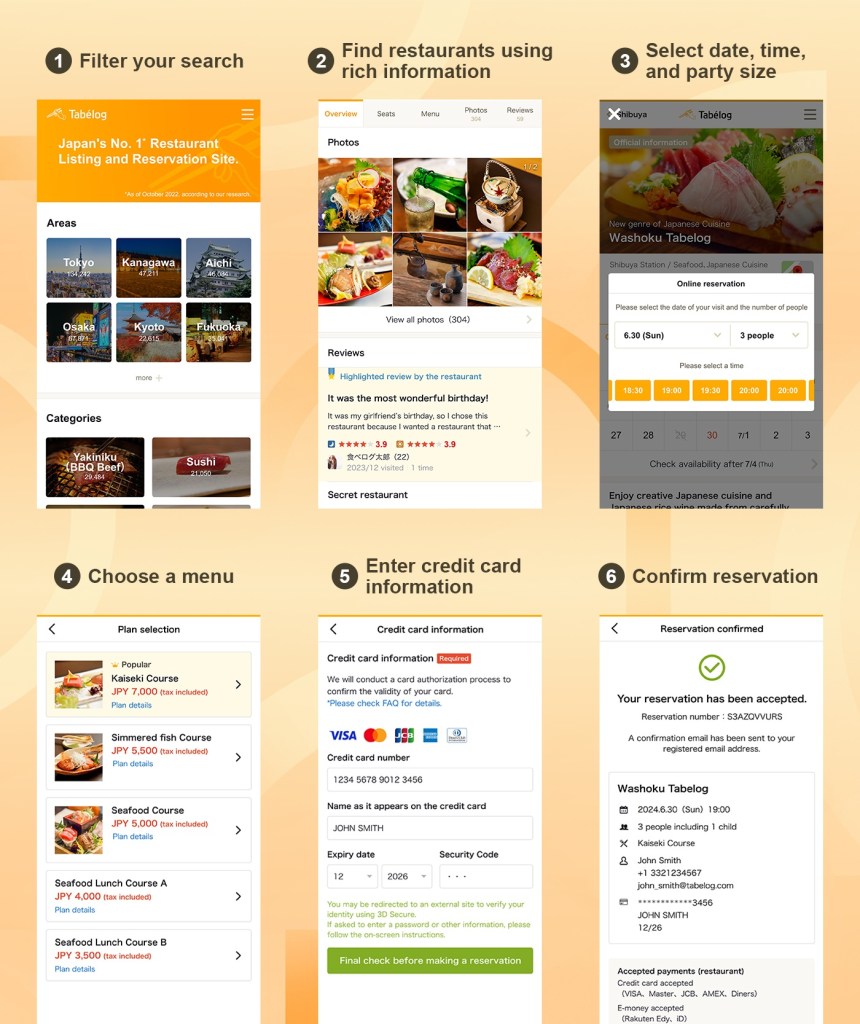
MARBURG, Germany, July 4 (Bernama-GLOBE NEWSWIRE) — Global biotechnology leader CSL Behring (ASX: CSL) today announced that two hemophilia B patients were treated with the gene therapy HEMGENIX® (etranacogene dezaparvovec) at Hemophilia Treatment Centers in France. This milestone achievement makes HEMGENIX® the first gene therapy administered as a treatment in a real-world setting for hemophilia B in Europe.
HEMGENIX® is the first one-time gene therapy approved in Europe for the treatment of adults with severe and moderately severe hemophilia B, an inherited bleeding disorder caused by the lack of Factor IX (a protein needed to produce blood clots to stop bleeding). It is used in adults without a history of Factor IX inhibitors.¹
Following European Commission approval, HEMGENIX® was the first ever therapy to be granted Direct Access in France², thus enabling the first patients to be treated in Europe outside of the clinical program.
Though effective, current therapies can be time intensive and require regular treatment that can have a substantial impact on a patient’s daily life.³ HEMGENIX® offers a one-time treatment, allowing people living with hemophilia B to produce their own Factor IX, which can lower the risk of bleeding.⁴
“Only a few decades ago, gene therapy for hemophilia was a distant concept, which has now become reality. Accordingly, the first two patients treated with HEMGENIX® since receiving European approval is a major accomplishment and a testament to the joint commitment of the hemophilia B community, as well as the access and reimbursement authorities, in bringing innovative therapies to patients,” said Dr Lutz Bonacker SVP and General Manager, CSL Behring Commercial Operations Europe. “This milestone has been made possible by the innovative Direct Access scheme adopted in France, allowing patients to benefit from early access to pioneering treatments. We are encouraged to see increasing access to gene therapies in European countries and are fully committed to ensuring that access to potentially life-changing treatment continues.”
HEMGENIX® was granted conditional marketing authorisation by the European Commission (EC) for the European Union and European Economic Area in February 2023, following approval from the U.S. Food and Drug Administration (FDA) in November 2022. It has also been approved by Health Canada, the United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA), Switzerland’s Swissmedic and Australia’s Therapeutic Goods Administration (TGA).
The multi-year clinical development of HEMGENIX® was led by uniQure and sponsorship of the clinical trials transitioned to CSL after it licensed global rights to commercialise the treatment.
About Hemophilia B
Hemophilia B is a life-threatening rare disease. People with the condition are particularly vulnerable to bleeds in their joints, muscles, and internal organs, leading to pain, swelling, and joint damage. Current treatments for moderate to severe hemophilia B include life-long prophylactic infusions of factor IX to temporarily replace or supplement low levels of the blood-clotting factor.
About HEMGENIX®
HEMGENIX® is a gene therapy that reduces the rate of abnormal bleeding in eligible people with hemophilia B by enabling the body to continuously produce factor IX, the deficient protein in hemophilia B. It uses AAV5, a non-infectious viral vector, called an adeno-associated virus (AAV). The AAV5 vector carries the Padua gene variant of Factor IX (FIX-Padua) to the target cells in the liver, generating factor IX proteins that are 5x-8x more active than normal. These genetic instructions remain in the target cells, but generally do not become a part of a person’s own DNA. Once delivered, the new genetic instructions allow the cellular machinery to produce stable levels of factor IX.
About the Pivotal HOPE-B Trial
The pivotal Phase III HOPE-B trial is an ongoing, multinational, open-label, single-arm study to evaluate the safety and efficacy of HEMGENIX®. Fifty-four adult hemophilia B patients classified as having moderately severe to severe hemophilia B and requiring prophylactic factor IX replacement therapy were enrolled in a prospective, six-month or longer observational period during which time they continued to use their current standard of care therapy to establish a baseline Annual Bleeding Rate (ABR). After the six-month lead-in period, patients received a single intravenous administration of HEMGENIX® at the 2×10^13 gc/kg dose. Patients were not excluded from the trial based on pre-existing neutralizing antibodies (NAbs) to AAV5.
A total of 54 patients received a single dose of HEMGENIX® in the pivotal trial, with 52 patients completing at least three years of follow-up. The primary endpoint in the pivotal HOPE-B study was ABR 52 weeks after achievement of stable factor IX expression (months 7 to 18) compared with the six-month lead-in period. For this endpoint, ABR was measured from month seven to month 18 after infusion, ensuring the observation period represented a steady-state factor IX transgene expression. Secondary endpoints included assessment of factor IX activity.
No serious treatment-related adverse reactions were reported. One death resulting from urosepsis and cardiogenic shock in a 77-year-old patient at 65 weeks following dosing was considered unrelated to treatment by investigators and the company sponsor. A serious adverse event of hepatocellular carcinoma was determined to be unrelated to treatment with HEMGENIX® by independent molecular tumour characterization and vector integration analysis. No inhibitors to factor IX were reported.
Long-term three-year data presented at the 17th Annual Congress of the European Association for Haemophilia and Allied Disorders (EAHAD) 2024 continue to reinforce the potential long-lasting efficacy and safety of HEMGENIX® and the ongoing benefit of this treatment for people living with hemophilia B.
About CSL
CSL (ASX:CSL; USOTC:CSLLY) is a global biotechnology company with a dynamic portfolio of lifesaving medicines, including those that treat hemophilia and immune deficiencies, vaccines to prevent influenza, and therapies in iron deficiency and nephrology. Since our start in 1916, we have been driven by our promise to save lives using the latest technologies. Today, CSL – including our three businesses: CSL Behring, CSL Seqirus and CSL Vifor – provides lifesaving products to patients in more than 100 countries and employs 32,000 people. Our unique combination of commercial strength, R&D focus and operational excellence enables us to identify, develop and deliver innovations so our patients can live life to the fullest. For inspiring stories about the promise of biotechnology, visit CSL.com/Vita. For more information about CSL, visit CSL.com.
Media Contacts
Stephanie Fuchs
Mobile: +49 151 584 388 60
Email: Stephanie.Fuchs@cslbehring.com
References
¹ European Medicines Agency. First Gene therapy to treat haemophilia B. Available at: https://www.ema.europa.eu/en/news/first-gene-therapy-treat-haemophilia-b. [Accessed May 2024].
² Republique Française. Légifrance: Article 62 of Law No. 2021-1754. Available at: https://www.legifrance.gouv.fr/jorf/id/JORFTEXT000048551003 [Accessed May 2024].
³ Leebeek, F & Miesbach, W. (2021) Gene therapy for haemophilia: a review on clinical benefit, limitations, and remaining issues. Blood. Vol 138, Issue 11. pp923-931.
⁴ Coppens M et al. Etranacogene dezaparvovec gene therapy for haemophilia B (HOPE-B): 24-month post-hoc efficacy and safety data from a single-arm, multicentre, phase 3 trial. The Lancet Haematology 2024; 11(4):E265-E275.
SOURCE: CSL Behring
–BERNAMA